Controversy Alert! Use of Aspirin for Primary Prevention
Steph’s Note: This week, we’re back with another data-filled post from Christian Gonzalez-Hernandez. (No, I’m not even kidding. He’s making all of us look bad with his insane level of productivity! He’s cranking out these posts during quarantine like there’s no tomorrow, and we love him for it!)
This time, he’s shifting his focus to the cardiology world and one of our oldest drugs: aspirin. If you thought you knew whether and when to add this seemingly simple OTC medication to a patient’s regimen, you may want to give this another look… Happy reading!
Controversy Alert!
Use of Aspirin For Primary Prevention of Atherosclerotic Cardiovascular Events
Imagine reading this headline:
Professor Oak’s been pretty busy adding the study of aspirin to his day to day. (Image)
“Researchers at Oak University in Pallet Town discovered that aspirin reduces the risk of cardiovascular events.“
And there you go. You are all hyped up to make the recommendation to add aspirin to all your patients - only to hear the following on the news just eight months later:
“Researchers at Misty University in Cerulean City found that using aspirin to reduce the risk of
cardiovascular events leads to an increase in bleeding.”
And so the story goes...
I bet all of us have seen similar headlines on the news, and they serve to teach us a lesson. One size does not fit all, especially when talking about primary prevention of atherosclerotic cardiovascular disease (ASCVD) with aspirin.
Spoiler alert 🚨: Primary prevention with aspirin is a pivotal component of many patients’ pharmacotherapy regimens, but not every person needs or may benefit from this. While the use of low-dose aspirin for the secondary prevention of ASCVD is widely accepted and implemented, the same cannot be said about its use for primary prevention.
So let’s dive into a literature review to find out when to use it - and when not to use it.
But first, let’s go back in time and see how the world’s most widely used drug came to be.
How Does Aspirin Work?
In ancient Egypt and Greece, willow bark was believed to cure aches and fevers. These days, we know those people actually weren’t crazy!
The active extract of willow bark is ‘salicin’, and its pure state is called salicylic acid. In the late 1800s, this product was used to treat rheumatic fever and arthritis-related pain.
Due to the high frequency of adverse effects, the search for a safer and more efficacious compound was on like Donkey Kong. Friedrich Bayer hoped to build on the success of salicylic acid, but it was actually Felix Hoffman who found a way to add an acetyl group in place of the hydroxyl group located on salicylic acid’s benzene ring.
For those of you who reeeeeally loved med chem…
Enter acetylsalicylic acid. *booming dramatic music*
But to avoid confusion between it and its predecessor salicylic acid, the name aspirin was given to the new compound.
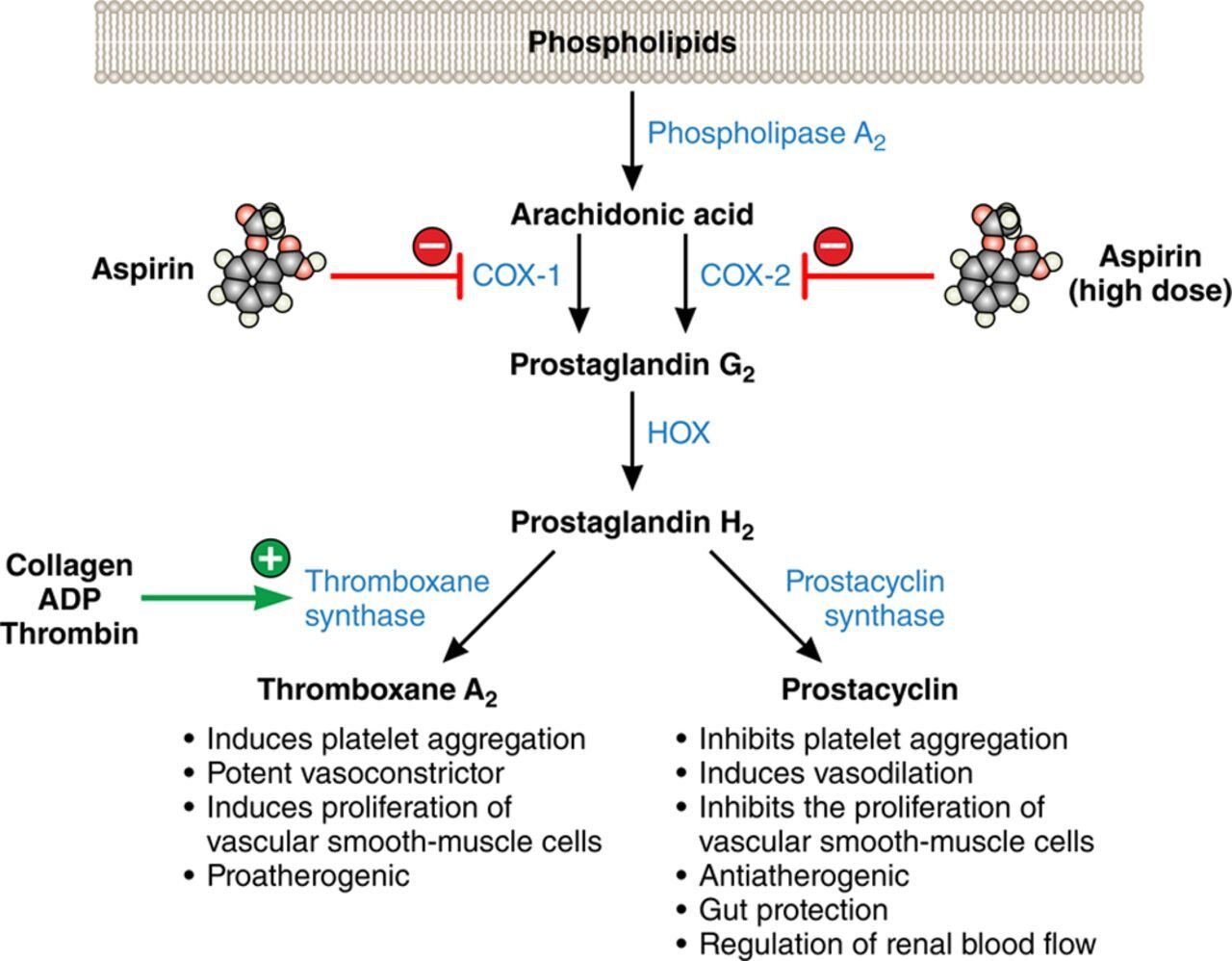
In certain situations, arachidonic acid is liberated from the cell phospholipid bilayer through the action of an enzyme called phospholipase 2a. Arachidonic acid is then metabolized via prostaglandin G synthase and prostaglandin H synthase (aka COX-1 and COX-2, respectively) to form prostaglandin G2 (PGG2) and prostaglandin H2 (PGH2). These are intermediaries in the formation of prostanoids like thromboxane and prostacyclin. Thromboxane has important functions in the aggregation of platelets, and it also has vasoactive properties.
For those visual folks:
(Image)
Acetylsalicylic acid, better known as aspirin, works in several pathways involving inflammation, hemostasis, thrombosis, and cell proliferation. In terms of cardiovascular protection, aspirin’s irreversible binding to COX-1 and COX-2 via covalent acetylation of critical serine residues in those enzymes leads to decreased production of thromboxane and decreased platelet aggregation.
Aspirin For Secondary Prevention of ASCVD
Before discussing controversial primary prevention, let’s talk about the less controversial topic of aspirin for secondary prevention of ASCVD.
It is well documented that the use of aspirin for secondary prevention of ASCVD is effective and widely implemented in practice. There is a huge amount of clinical data supporting its use in this specific setting.
In 2012, the Antithrombotic Trialists’ Collaboration performed a meta-analysis of over 280 studies involving 135,000 patients with prior history of myocardial infarction (MI), stroke, peripheral artery disease (PAD), etc. The primary outcome was the incidence of ‘serious cardiovascular events’, which was defined as: non-fatal myocardial infarction, non-fatal stroke, or vascular death.
Antiplatelet therapy reduced the composite outcome by about one quarter. There was no difference in the primary outcome when lower aspirin doses were compared to higher doses of aspirin. When compared with other antiplatelets, aspirin was non-inferior.
Another meta-analysis performed in 2008 evaluated trials of low-dose aspirin in stable cardiovascular disease. Six trials were included (4 for stroke/TIA, 1 for MI, 1 for stable angina) covering a total of 9,853 patients. The main outcome was a composite of cardiovascular events: nonfatal MI, nonfatal stroke, cardiovascular death.
Aspirin was associated with a 21% decrease in the main outcome (95% CI 0.72-0.88; P=0.01), with a number needed to treat of 30 based on an absolute risk reduction of 3.3%. This means it only took giving aspirin to 30 patients to prevent 1 adverse outcome. Not too shabby.
Major bleeding was more common in the aspirin users with an absolute risk increase of 0.9% (95% CI 1.41-3.35; P=0.01). The number needed to harm was 111, meaning it took treating 111 patients with aspiring to see 1 bleeding event.
All in all, during an average follow-up of 33 months, if 1000 patients are treated with aspirin, 33 cardiovascular events would be prevented while only 9 bleeding events would occur. This suggests that the benefits of aspirin for secondary prevention outweigh the risks.
(Note: those numbers are calculated by dividing 1000 by the NNT or NNH. They give us some perspective of how the NNT and NNH stack up against each other.)
So now that we’ve established that aspiring for secondary prevention is good stuff, let’s head for the controversy.
The REAL Controversy: Aspirin For Primary Prevention of ASCVD
When talking about aspirin use for secondary prevention of cardiovascular events, there’s a consensus. We cannot say the same when speaking about primary prevention, for which there’s a fine line between the risks and benefits. Even amongst guidelines, there is controversy regarding the use of aspirin for primary prevention!
Cardiovascular risk levels according to the 2019 ESC Guidelines.
b = proteinuria, renal impairment defined as eGFR <30 mL/min/1.73m2, left ventricular hypertrophy, or retinopathy
c = age, hypertension, dyslipidemia, smoking, or obesity
(Image)
The 2012 American College of Chest Physicians Guidelines on Primary and Secondary Prevention of Cardiovascular Diseases recommend (or suggest, to be more specific) low-dose aspirin (75–100 mg daily) in patients aged >50 years (Class 2B recommendation).
The 2019 ESC/EASD Guidelines do not recommend the use of aspirin for primary prevention, even in those patients with moderate cardiovascular risk. Additionally, they only provide a low-confidence recommendation for antiplatelet therapy in patients with diabetes mellitus with high or very high cardiovascular risk (IIb).
The 2020 ADA Standards of Care in Diabetes state, “Aspirin therapy (75–162 mg/day) may be considered as a primary prevention strategy in those with diabetes who are at increased cardiovascular risk, after a comprehensive discussion with the patient on the benefits versus the comparable increased risk of bleeding. (A)”
And even though the exact same text is used in the 2019 ADA Guidelines, the confidence level in the 2019 version was a Class C instead of the above Class A.
Big change in confidence over the course of one year. Huh. Why might that be…
Finally, the 2019 ACC/AHA Guideline on Primary Prevention stratifies their recommendations based on ASCVD and bleeding risks. This document offers 3 recommendations for the use of aspirin for primary prevention:
Low-dose aspirin (75–100 mg PO daily) might be considered for the primary prevention of ASCVD among selected adults aged 40–70 years who are at higher risk of ASCVD but not at increased risk of bleeding (IIb).
Low-dose aspirin (75–100 mg PO daily) should not be administered on a routine basis for the primary prevention of ASCVD among adults aged >70 years.
Low-dose aspirin (75–100 mg PO daily) should not be administered for the primary prevention of ASCVD among adults of any age who are at increased risk of bleeding.
As you can see, there is some discordance in the published recommendations, and assessing the current literature to identify which populations may benefit can be a difficult task. So, being (future) pharmacists who love evidence-based medicine, let’s dive into the available literature to see how these recommendations came to be!
A Brief History of Trials Studying Aspirin for ASCVD Primary Prevention
The first trial that attempted to assess the efficacy and safety of aspirin for primary prevention of cardiovascular events was the 1988 British Doctor’s Study (BDS). This six-year study evaluated whether aspirin 500 mg PO daily (regular release, btw) could reduce the incidence of, and mortality from, stroke, MI, or other vascular conditions in 5139 otherwise healthy British doctors.
Over 3400 doctors were allocated to the aspirin group, while 1710 were instructed to avoid aspirin. Six hundred and seventy participants stopped using aspirin during the first year, with an additional 678 people stopping aspirin over the course of the subsequent 5 years (about 125 doctors per year). Bleeding and dyspepsia were the main reasons that led to aspirin discontinuation.
(Not exactly a shocker with what we know now… 500mg of non-enteric coated aspirin every day is pretty substantial.)
The use of aspirin in this trial was not associated with lower incidences of stroke or MI. Deaths related to MI or stroke were not significantly reduced by the use of aspirin either. Peptic ulcer disease and non-fatal bleeding rates were higher in the group taking aspirin. Not exactly a whopping endorsement for use of aspirin, but there are some specific things to note about this study…
First, the dose of aspirin was 500 mg, which is a ~5x our current low-dose 75-100mg daily strategy.
Second, they used the non-enteric coated formulation, which is more harsh on the stomach than the enteric coated.
Third, the population was comprised of all physicians. Likely not the most generalizable in terms of baseline health and event rates.
A few years later in 1989, a similar study was performed in America. The Physician’s Health Study included 22,071 physicians who were randomized in 2:2 factorial design to receive one of the following regimens: 325 mg of aspirin every other day and beta carotene, aspirin and placebo, placebo and beta carotene, and all placebo.
All in all, 11,037 people received aspirin, and 11,034 patients received placebo. They were followed for an average of 60 months.
Aspirin use was associated with a reduction in MI (RR 0.56, 95% CI 0.45 - 0.70; P < 0.0001). There was no difference observed in stroke. Using a combined endpoint of cardiovascular death, MI, and stroke, there were 307 events in the aspirin group compared to 370 events in the placebo group (95% CI 0.70 - 0.96; P = 0.01). A subgroup analysis showed older and obese patients seemed to benefit the most from aspirin. As with other studies, bleeding was more common in the aspirin groups.
Basically, what the aforementioned studies suggest is the following:
Aspirin may or may not be beneficial for the primary prevention of cardiovascular events.
Older patients seemed to benefit the most from aspirin.
This benefit may come at the cost of bleeding complications.
In healthy, young patients, this potential cardiovascular benefit may not be worth the risk of the associated bleeding complications.
For additional context…
According to the 1989 US PHS trial, in patients 40 to 49 years old, we need to treat 1,000 patients to prevent 1 MI. In patients aged 60-69 years, we only need to treat 46 patients to prevent 1 MI. That number goes down further to 30 in patients 70 or older! Huge difference when stratifying by age.
Nonetheless, when we look at it in general without taking age into consideration, the number needed to treat is 175.
On the other hand, for every 7 patients treated with aspirin, 1 will have a bleeding event. Not so great of a number needed to harm.
So if 1000 patients are treated with aspirin, 6 primary outcome events will be prevented, while 151 bleeding events will be caused…of which 11 will be melena, 2 will be non-GI bleeding, and the others will be less serious events like easy bruising and nosebleeds.
Yikes…those numbers… (Image)
In 2016, the US Preventive Services Task Force released a systematic review of 11 primary prevention trials of aspirin. Two good-quality and 9 fair-quality randomized, controlled trials were identified. Aspirin reduced the risk for non-fatal MI (RR 0.78, 95% CI 0.71 to 0.87) but not non-fatal stroke. Aspirin showed little or no benefit for all-cause or cardiovascular mortality.
In the end, the authors concluded that the beneficial effects of aspirin for the primary prevention of ASCVD are modest, and they occur at doses of 100 mg or less per day. Once again, the patients who benefited the most were older patients.
Despite all of the studies involved in this systematic review, not a single one was able to identify a benefit in terms of reduction in non-fatal stroke and mortality. And only 4 of the 11 studies demonstrated significant reductions in the rates of non-fatal MI among low-dose aspirin users. Bleeding episodes were again more common with aspirin.
This is such a good example of the importance of balancing ‘risk vs. benefit’ when treating a particular patient. So how do we identify those patients who WILL benefit from aspirin primary prevention?
Based on this, subsequent studies were geared towards identifying those specific subgroups who are at an increased risk of MI, stroke, or cardiovascular deaths and may derive the most benefit from low-dose aspirin. As such, we are going to look at studies focused on patients with diabetes (ASCEND), older age (ASPREE), or a combination of risk factors (ARRIVE).
Recent Subgroup Studies of Aspirin for ASCVD Primary Prevention
2018 was a big year for aspirin in the setting of primary prevention! The ARRIVE, ASPREE, and ASCEND trials were all published in late 2018, providing us with a lot of insight into the role of aspirin in primary prevention.
The ARRIVE Trial
The ARRIVE trial was a randomized, double-blind, placebo-controlled, multicenter study that assessed the role of aspirin for primary prevention in male patients at least 55 years old who had 2-4 risk factors for ASCVD. Females who were at least 60 years old and had 3+ risk factors were also included. Risk factors were the following:
High cholesterol (total cholesterol >200 mg/dL or LDL >130 mg/dL for men; total cholesterol >240 mg/dL or LDL >160 mg/dL for women)
Currently smoking or smoked within the last 12 months
Low HDL cholesterol (<40 mg/dL)
High blood pressure (systolic blood pressure >140 mmHg) or receiving pharmacotherapy for high blood pressure
Positive family history of cardiovascular heart disease.
Generally speaking, having 2-3 of these risk factors would confer an estimated 10-20% 10-year ASCVD risk. These authors reported 10-year ASCVD risks of 17.3% and 17.4% in the aspirin and placebo groups, respectively, so pretty well aligned there. People with diabetes were excluded from this trial because, according to the authors, these patients are at an increased risk of cardiovascular complications.
6270 patients were randomized to receive aspirin, while 6276 patients received placebo. The main outcome was a composite of confirmed MI, stroke, cardiovascular death, unstable angina, or transient ischemic attack (TIA). No difference was seen between groups for the primary composite outcome (269 events in the aspirin group (4·29%) vs. 281 events in placebo group (4·48%), 95% CI 0·81–1·13; p=0·6038). Additionally, no difference was seen in any of the individual components of the composite outcome (not even in MI as was the case in previous studies).
Gastrointestinal bleeding events occurred in 61 (0·97%) patients in the aspirin group and 29 (0.46%) patients in the placebo group (95% CI 1·36–3·28; p=0·0007). Subgroup analyses according to sex, age, smoking status, BMI, and ASCVD risk scores were performed, but no benefit was seen to justify the use of aspirin.
Sometimes how it feels when talking about statins versus other classes of medications… (Image)
Hard to know why aspirin was so blah in this study, but perhaps it was an effect of having more treatment options in recent times, including the statins (aka the kings of primary prevention in the 21st century).
The ASPREE Trial
The 2018 ASPREE trial was another big hit for aspirin in primary prevention. And not a hit in a good way. It basically added insult to injury.
In the ASPREE trial, not only did aspirin not confer any additional benefits, but it also suggested that in the elderly, the use of aspirin for primary prevention might even be harmful!
Community-dwelling patients at least 70 years old who did not have cardiovascular disease, dementia, or disability participated in this study. All-cause mortality was higher in the group taking aspirin, with the major driver being cancer-related deaths. (Interestingly, cancer-related deaths were significantly higher in the aspirin group, but the use of aspirin as chemoprophylaxis against some types of cancers is in and of itself a lengthy and controversial topic!).
No significant difference was seen in cardiovascular-related deaths in this trial. Furthermore, aspirin did not provide any benefits with regards to the composite primary outcome (or its individual components) of all-cause mortality, dementia, and physical disability. This is a break from the usual primary outcomes in these types of studies, but it’s also a pretty reflective endpoint if you’re considering what matters to older adults.
Bleeding was another story. Major hemorrhagic events were significantly higher among aspirin users (361 vs. 265, HR 1.38, 95% CI 1.18–1.62; P<0.001).
It is likely that the ACC/AHA made the recommendation to not use aspirin in patients who are 70 years or older in their 2019 Primary Prevention Guidelines based on the results of this trial. It should be noted that ASPREE was comprised of a predominately white population. Latino and Black patients made up only about 8% of the total trial population, so generalizing the study results to other ethnicities may be a little premature…even though subgroup analyses indicated that the effects of aspirin did not differ across the different subgroups evaluated in this study.
But were there enough people in those subgroups to detect a difference?
The ASCEND Trial
The last of the trials released in 2018 was the ASCEND trial, which evaluated the use of aspirin in 15,480 patients with diabetes, were at least 40 years old, and did not have evidence of cardiovascular disease. The primary outcome was a composite of the first recurrence of MI, stroke, TIA, or death due to cardiovascular causes.
The primary safety outcome was a composite of major bleeding events (aka intracranial hemorrhage, sight-threatening bleeding event in the eye, gastrointestinal bleeding, or any other serious bleeding event that resulted in hospitalization, transfusion, or death).
Aspirin was associated with a reduction in the composite primary outcome (658 [8.5%] vs. 743 [9.6%]; rate ratio 0.88, 95% CI 0.79 to 0.97; P = 0.01). When looking at the individual components of the primary efficacy outcome, none of them showed a significant difference.
Major bleeding was higher in the aspirin group, which is consistent with other trials (314 [4.1%] vs. 245 [3.2%]; rate ratio 1.29, 95% CI 1.09 to 1.52; P = 0.003). The number needed to treat was 91, while the number needed to harm was 111.
To put this another way… During an average of 7.4 years, if 1000 diabetic adults over age 40 are treated with aspirin for primary prevention of ASCVD, 9 bleeding events will occur, while 11 cardiovascular events will be prevented.
Hard to make a determination isn’t it?
Basically, we have the risk of bleeding almost evenly counterbalancing the benefits of aspirin!
This prompted the ADA to recommend, “Aspirin therapy (75–162 mg/day) may be considered as a primary prevention strategy in those with diabetes who are at increased cardiovascular risk, after a comprehensive discussion with the patient on the benefits versus the comparable increased risk of bleeding.”
Lots of “mays” and “ifs” in there… So it is not a definitive recommendation, but a conditional one that requires an in-depth discussion on an individualized basis.
Reading between the Lines on Aspirin for Primary Prevention
While evidence suggests that lower doses of aspirin produce similar cardioprotective effects to higher doses, earlier trials often used higher doses (and riskier formulations) of aspirin. So there were increased bleeding events. Not exactly a huge surprise.
These trials mostly showed reductions in the rates of MI, while not producing significant effects in the rates of stroke and cardiovascular death. Later, but pre-2018, trials that used lower doses of aspirin noticed inconsistent effects in the rates of cardioprotection. Before the big 3 trials in 2018 (ARRIVE, ASPREE, and ASCEND), only the 1998 HOT trial (aspirin dose 75 mg), the 2014 JPPP trial (aspirin dose 100 mg), the USPHS trial (aspirin dose 162 mg), and the 1998 TPT trial (aspirin dose 75 mg) were able to detect lower rates of MI among aspirin users.
Anyways, when taking all aspirin for primary prevention trials into account, the overall tendency favors the use of aspirin for cardiovascular protection…but this is potentially at the cost of bleeding. So it’s possible that the benefits from aspirin may be counterbalanced by its risks.
It is also worth noting that all post-2000 trials of aspirin for primary prevention did not detect any significant benefits. Even though the first FDA-approved statin medication was lovastatin in 1987, it wasn’t until the late 1990s/early 2000s that statins became a mainstay for the primary prevention of ASCVD. Could the introduction of statins be why the later trials didn’t find a significant difference with aspirin? Like aspirin is irrelevant in the world of statins?
Perhaps some of the variability in aspirin’s demonstrated effects also has to do with individual patient factors and comorbidity management. One 2009 meta-analysis of 6 trials with 95,000 participants tried to tease out benefits of aspirin primary prevention based on individual patient characteristics. Based on the Forest plot below, you can see it wasn’t too awfully discerning for which groups benefit most…and some of the boxes even seem contradictory to what later trials demonstrated.
Not particularly revealing, is it… (Image)
Now about those comorbidities…
In older trials, it’s entirely possible blood pressure control was suboptimal, lipid lowering therapies were rare, and smoking was more common than recent years, so perhaps those included patients were at a higher risk for ASCVD. But now, with better comorbidity management, perhaps the addition of aspirin just isn’t that big of a deal.
Hard to say for sure.
But perhaps newer trials like the ASCEND, ASPREE, and ARRIVE provide better insight into the impact (or lack thereof) of aspirin in the context of current times, when a significant proportion of the population is using statin therapy. For each of those trials, 34%, 43%, and 75% of patients, respectively, were on a statin.
The authors of the ARRIVE trial even state, “The targeted estimated risk for enrollment in ARRIVE was achieved, but the observed event rate was considerably less than anticipated.” They attributed this to, “Patients are being treated for their risk factors to lower the risk of the development of disease. Better management of blood pressure, dyslipidemia, and other risk factors is likely to lower the risk of developing disease.”
So maybe we just need to do a better job of managing those comorbidities and other risk factors rather than risking aspirin.
But what about those patients who are at high risk of ASCVD? Does the risk/benefit ratio tip more towards benefit with aspirin in this group?
Again, hard to say - especially since determining who is “high risk” isn’t an exact science.
Better tools for identifying patients at exceptionally high risk of developing ASCVD may be warranted. The ARRIVE trial tried to assess this, but it ended up recruiting a patient pool with an average ACC/AHA 10-year risk of ASCVD of 17%, which may be considered as moderate. Perhaps performing studies in patients with a 10-year ASCVD risk of >20% or even >25% may provide better insight into whether “high risk” patients may benefit from aspirin primary prevention.
Another important factor to consider is that patients at higher risk of ASCVD were also at higher risk of bleeding with aspirin, as was seen in the meta-analysis performed in 2008 (see below).
MCE = major cardiovascular event; MEB = major extra cranial bleed
Finding ways to mitigate the risk of bleeding with aspirin in order to reap its potential benefits could be another area to investigate. The COMPASS trial assessed the use of pantoprazole in combination with low-dose anticoagulation (rivaroxaban 2.5 mg twice daily) with or without low-dose aspirin. Pantoprazole significantly reduced gastrointestinal bleeding, but there are some factors that need to be considered before we all jump on the proton pump inhibitor (PPI) + aspirin train in ASCVD primary prevention:
PPIs are a whole controversial topic in and of themselves due to the increased incidence of C. diff infections (and even reports of increased multi-drug resistance (MDR) infections).
This study assessed the use of rivaroxaban in the context of stable cardiovascular disease and not primary prevention.
This study assessed the use of rivaroxaban with or without aspirin, meaning patients were more anticoagulated than with aspirin alone.
This trial does not provide definitive evidence supporting the use of PPIs to counterbalance the risk of bleeding with aspirin in primary prevention. But it may serve to highlight the need for further studies about strategies to reduce the risk of GI bleeding with aspirin.
Perhaps trials evaluating the use of PPIs or H-2 receptor antagonists (H2RAs) in the setting of aspirin primary prevention in high-risk or very high-risk patients may provide better ideas of how to counterbalance the risk of bleeding in these patients.
And finally, we have heard countless times the need to balance potential benefits with risks, and guideline recommendations echo this sentiment. The 2020 ADA Standards of Care Guidelines for Diabetes state, “Aspirin therapy (75–162 mg/day) may be considered as a primary prevention strategy in those with diabetes who are at increased cardiovascular risk, after a comprehensive discussion with the patient on the benefits versus the comparable increased risk of bleeding.” The 2019 ACC/AHA Guidelines on Primary Prevention recommend, “Low-dose aspirin (75–100 mg orally daily) might be considered for the primary prevention of ASCVD among selected adults aged 40–70 years who are at higher risk of ASCVD but not at increased risk of bleeding.”
But what exactly qualifies as “increased risk of bleeding” and “increased” or “higher risk of ASCVD”?
This is what makes this topic such a controversy!
The ACC/AHA Guidelines provide a list of conditions that may increase bleeding risk, including:
History of previous GI bleeding, peptic ulcer disease, or bleeding at other sites
Age >70 years
Thrombocytopenia or coagulopathy
Chronic kidney disease (CKD)
Concurrent use of other medications that increase bleeding risk, such as NSAIDs, steroids, direct oral anticoagulants, and warfarin
This list might be useful for determining which patients may NOT be good candidates for aspirin primary prevention… But it doesn’t really help us figure out who would be good candidates. And it doesn’t help us figure out who has a high risk of ASCVD.
The ESC/EAS 2019 Guidelines provide some information that may not necessarily be perfect. But at least it’s some guidance! According to these guidelines, high risk ASCVD patients have had…
Diabetes for 10 or more years without target organ damage (defined as proteinuria, CKD stage IV, retinopathy or left ventricular hypertrophy), PLUS
One additional risk factor (i.e., age, hypertension, dyslipidemia, smoking, and obesity).
Very-high risk patients are defined as patients with…
Diabetes, PLUS
Established cardiovascular disease or target organ damage, OR
Three or more risk factors, OR
Type 1 diabetes onset at least 20 years ago.
Until new data emerges on the use of aspirin for primary prevention in patients with an ASCVD of 20% or more as well as strategies to reduce the risk of bleeding, the 2019 ESC/EAD guidelines provide at least some clear thresholds for defining high or very high-risk of ASCVD. It’s still not a fully comprehensive list, and it certainly doesn’t tell us for sure who will absolutely derive benefit from aspirin primary prevention. But it’s something perhaps a little more concrete!
In summary, the use of aspirin for primary prevention is not a one size fits all strategy. It requires evaluation on a case by case basis to determine if it will be both useful AND safe.