NICU Series Part 3: Necrotizing Enterocolitis (NEC) in Premature Neonates
Brandon’s Note: We’re coming in hot with the third installment of our NICU series with NEC in Premature Neonates. As with the first post and second post in the series, this one was written by the phenomenal Courtney Howell, PharmD. Fun fact: Courtney is also now an OFFICIAL tl;dr team member. You can reach her at courtney@tldrpharmacy.com. Now, let’s sit back and absorb the pediatric pharmacy knowledge that’s coming to us. Take it away, Courtney!
And BTW…we’ve got a super handy Pediatric Pharmacy Pocket Guide that gives you the ins and outs of all things peds. It’s an absolute life-saver…especially if you only occasionally come across pediatric patients in your practice. Check it out here!
I realized I’ve done a Spongebob meme in every article, so I can’t stop now.
It’s that time again! NECxt up (see what I did there?) in our NICU miniseries is a common gastrointestinal emergency in premature neonates: Necrotizing Enterocolitis (NEC).
Again, I will be using some of the age and weight-related abbreviations from the respiratory article, so be sure to take a look at that before you read on.
Am I just trying to get you to read my other articles? Maybe, but I’ll never tell…
*insert evil laughter here*
What is Necrotizing Enterocolitis (NEC)?
I think we can all agree that anything with the word “necrotizing” in it is, well…not good. Let’s use our medical terminology skills and break this down, shall we?
Necrotizing = causing or accompanied by necrosis (i.e. death of cells in an organ or tissue).
Entero = relating to the intestines.
Colitis = inflammation of the large intestine.
So, NEC is quite literally the inflammation and death of cells in the intestinal tract. The cause of NEC is not well-defined but is likely due to an immature digestive system in premature neonates. The structural barrier doesn’t mature until around 26 weeks, so babies born before or shortly after this time may not have digestive systems capable of keeping harmful bacteria out when inflammation (the “-itis” in enterocolitis) is present.
Inadequate blood flow to the intestines has also been implicated as a potential cause of NEC (shout out to our PDA article!), but its role is not well-established. Full-term neonates can develop NEC, too — although it is almost always due to underlying illness or comorbidities. In all cases, NEC should be considered a medical and surgical emergency as it can be life-threatening if not treated promptly.
How Does NEC Present?
Ahhhh, the ‘70s. A time when bell bottoms were cool, disco was all the rage, and everyone and their mother had a hideous orange sofa in the living room. This was also a time when NEC was still very poorly understood, and there was no real consensus on diagnostic criteria or treatment (this is still the case today, but our understanding of the disease and how to treat it is much better than it was fifty years ago).
Over in St. Louis circa 1978, Bell et. al. proposed the first classification system for NEC. This classification system broke NEC down into three stages:
Stage I (Suspect)
Stage II (Definite)
Stage III (Advanced)
Modified Bell's Staging Criteria were introduced by Walsh and Kliegman in 1986. These expanded the original three Bell stages into six and incorporated treatment options. You can view the full modified criteria by clicking the link above. If you want the quick version, I’ve summarized the table below (this is tl;dr, after all!).
Each of the three stages is split into two. A baby in stage I (suspected NEC) will have trouble maintaining a normal body temperature, be lethargic, and experience A’s and B’s (that’s what the cool kids call “apnea and bradycardia”). They may show signs of feeding intolerance (increased gastric residuals, mild abdominal distension, and vomiting).
Patients in all stages should be made NPO to allow the bowel to rest and heal. Total parenteral nutrition (TPN) will be continued until feeds are resumed, the timing of which will vary based on the clinical course.
The major difference between stage IA and IB is the blood content in the stool. Occult blood may be present in stage IA, but the stool will be grossly bloody in stage IB. Imaging can appear normal, or the intestines may be slightly dilated.
All babies with suspected or confirmed NEC will be given an NPO (“nil per os” meaning “nothing by mouth") order and babies in stage I are recommended to receive antibiotics for 3 days to prevent systemic infection.
Stage II indicates definite NEC. The severity of illness is defined as “mild” for Stage IIA and “moderate” for Stage IIB.
Stage IIA is pretty similar to stage I, but these babies will also have absent bowel sounds and some abdominal tenderness. Imaging may reveal the presence of gas within the wall of the small or large intestine. A longer antibiotic duration of 7-10 days is recommended for these cases.
Patients in Stage IIB will also have mild metabolic acidosis and low platelets. There will be definite abdominal tenderness and gas in the portal vein, and fluid in the abdomen may be present on imaging. An even longer antibiotic duration of 14 days is recommended. They’ll also usually get sodium bicarbonate to correct the acidosis.
In Stage III (advanced NEC), the major difference between stages is whether the bowel is intact (IIIA) or perforated (IIIB). These babies will have hypotension, respiratory acidosis, disseminated intravascular coagulation (DIC), and low neutrophils. They’ll also have marked abdominal tenderness and distention.
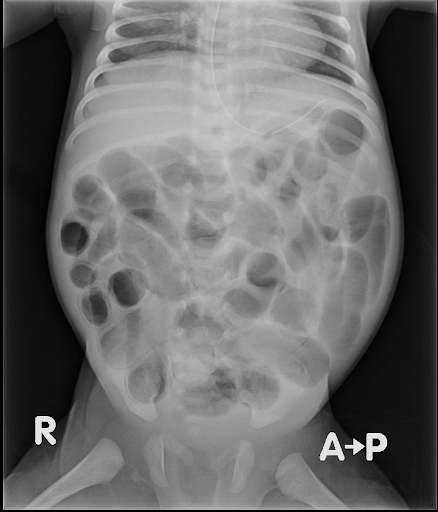
Imaging of a patient with NEC revealing abdominal distension and air in the bowel (pneumatosis intestinalis). image
Look at a CT of these poor kiddos, and you’ll see fluid and gas in the abdomen. The antibiotic duration for stage III is typically 14 days, but additional measures may be required (i.e. increased fluids, inotropic agents, mechanical ventilation, and paracentesis). Surgical intervention is required since a perforated bowel is a defining factor for Stage IIIB.
Both the original and modified Bell’s Staging Criteria are still widely utilized today, although there are limitations to their use. Many definitions of NEC exist, so it is important to always refer to your institution for guidance.
The image from an abdominal X-ray of a patient with NEC is something my pharmacotherapy professors would call “PharmD positive,” meaning something is so obviously wrong that even a pharmacist with no experience reading imaging can tell what is happening.
I will spare you photos of the clinical presentation of NEC as they can be pretty gnarly, but on the left is an abdominal X-ray of a patient with NEC. Google at your own risk.
NEC Treatment
Much like the definition, the treatment of NEC varies from institution to institution. Unfortunately, there are no consensus treatment guidelines, which can lead to much variability in practice.
A quality improvement project conducted for the level IV NICU at Massachusetts General Hospital (MGH) highlights this issue and provides a guideline based on available literature. I think the guideline is a pretty solid representation of “usual” practice, so I’ll summarize it here.
The first thing we should do when NEC is suspected is obtain bloodwork. The MGH guideline recommends blood cultures (prior to antibiotic administration, of course), a complete blood cell count (CBC) with differential, basic metabolic panel (BMP), and venous blood gas (VBG).
We also want an abdominal X-ray and any other recommended imaging. As stated above, all patients with suspected NEC should be made NPO to allow the bowel to rest, and a Replogle Tube may be placed to allow for gastric decompression.
Initiation of TPN may be warranted if the patient is expected to be NPO for an extended period (usually more than 48 hours). Famotidine should be avoided in the TPN as histamine type-2 receptor antagonists (H2RAs) have been associated with higher rates of NEC.
Antibiotics on their way to destroy bacteria and prevent systemic infection. The Twilight baseball scene was the cultural reset we all needed. I will die on this hill. image
Antimicrobial selection can be tricky as there is no single pathogen or known group of pathogens that cause NEC.
We are dealing with the bowel, so we need gram-positive, gram-negative, and anaerobic coverage. Because of this, empiric treatment is broad and can be narrowed once cultures and sensitivities come back. The antibiotic of choice in the MGH guideline is piperacillin/tazobactam, although many other combinations have been reported in the literature.
A joint guideline from the Surgical Infection Society (SIS) and Infectious Diseases Society of America (IDSA) published in 2010 recommends:
Ampicillin + gentamicin + metronidazole, OR
Ampicillin + cefotaxime + metronidazole, OR
Meropenem
It also recommends vancomycin instead of ampicillin for suspected methicillin-resistant Staphylococcus aureus (MRSA) or ampicillin-resistant enterococcal infection.
Fungal infections may also occur, in which case fluconazole or amphotericin B are recommended. There aren’t good studies suggesting one regimen over another, so it is important to refer to your institution for guidance.
Feeds will be restarted, and TPN will be weaned upon recovery at the discretion of the neonatologist. Human breast milk or pasteurized donor milk (PDM) are preferred when restarting feeds but can be switched to formula once tolerating full feeds based on parental preference. Surgery may be indicated for advanced cases.
NEC Prevention
One of the best defenses we have against NEC is human milk. Many studies have demonstrated a reduced incidence of NEC with human milk feeding, and a 2022 Policy Statement from the American Academy of Pediatrics states that PDM is recommended when a mother’s milk is unavailable or contraindicated (a little plug for any of you nursing mamas out there to go donate if you have extra milk!).
TPN is generally initiated for Very Low Birth Weight (VLBW) and Extremely Low Birth Weight (ELBW) infants, as their GI tracts are not equipped to handle full enteral feeds. The American Society for Parenteral and Enteral Nutrition (ASPEN) recommends minimal enteral nutrition (also called “trophic feeds”) be initiated in infants ≥ 1000 g within the first two days of life. These feeds (generally 20 mL/kg/day or less) do not provide nutrition but rather serve to stimulate the gut and have been shown to prevent NEC.
Another strategy increasing in popularity is giving probiotics. Like a teenager, a baby’s gut microbiome is young and impressionable. Most of the baby’s gut flora comes from the mother through delivery and breast milk feeding.
Healthy colonization of the gut can be disrupted by many factors, especially in premature neonates:
Maternal disease
Complications resulting in exposure to microbes in utero or during delivery
Delivery by C-section
Not receiving breast milk
Early antibiotic use
Exposure to hospital microbes during NICU admissions
Delays in enteral feeding due to prematurity
Despite an increase in probiotic use for the prevention of NEC, the AAP (and the European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) and Canadian Paediatric Society (CPS), for that matter) has issued a statement advising AGAINST the routine use of probiotics in preterm infants, especially in those < 1000 g. As of this writing, there is no data supporting probiotic use in this population. No clinical trial has shown a reduction of NEC in infants at the highest risk of complications, and the long-term safety is unknown.
There have also been reports of probiotic-associated sepsis that stems from the colonizing strain in the probiotic or a contaminant introduced during manufacturing. Currently no pharmaceutical-grade products are available, so we are relying on products not regulated by the Food and Drug Administration (FDA). An ongoing phase III randomized controlled trial (RCT) known as The Connection Study aims to solve this problem. A pharmaceutical-grade human bacterial strain naturally occurring in breast milk, Lactobacillus reuteri, is being evaluated in preterm infants with a birth weight of 500 to 1500 g and compared to placebo for safety and efficacy in preventing NEC. The trial is expected to be completed at the end of this year.
If you recall from the respiratory article, administering antenatal corticosteroids speeds up the maturation of fetal cells and prevents many complications of prematurity, including NEC. Wow, what a great catch-all article. You should really give that one a read!
The tl;dr of NEC
A second Spongebob meme for your viewing pleasure.
Multiple factors and pathogens contribute to the development of NEC, so finding optimal prevention and treatment strategies is like finding a working McDonald’s ice cream machine -- it’s really, really hard.
We do know that NEC should be considered a medical and surgical emergency as it can have dire consequences if left untreated. Our good friend Bell proposed the first NEC staging criteria, and our buddies Walsh and Kliegman modified those criteria into six stages still widely used for diagnosing NEC.
All patients with suspected or confirmed NEC should be made NPO, and broad-spectrum empiric antibiotics should be initiated after cultures are drawn to prevent bacterial translocation and systemic infection. Additional treatment interventions and the duration of antibiotics will depend on the severity of the disease and the clinical course.
Surgery may be indicated for advanced cases. Proven prevention measures for NEC include antenatal corticosteroid administration and early enteral feeding, preferably with the mother’s breast milk.
Probiotics have been used, but further research regarding their safety and efficacy is needed before they can be universally recommended for NEC prevention.
And there you have it. You are now a certified NECxpert! Okay, last cheesy pun, I promise. In this article, at least.