Ice, Ice Baby: A Dive into Targeted Temperature Management (TTM)
Steph’s Note: He’s baaaack! It’s our favorite journal club aficionado, Dr. Christian González-Hernández, PharmD, BCPS. You likely remember him from his previous data-driven, clinical trial-palooza posts on heart failure and secondary stroke prevention, aspirin for cardiovascular primary prevention, and sepsis, and if you don’t, you should hop over to those ASAP. Dr. González-Hernández graduated from Nova Southeastern University in May 2020. He completed a PGY1 residency at Broward Health Medical Center and is currently working as a critical care/cardiology clinical pharmacy specialist. When not watching anime or sports, he is reading clinical trials for fun, just to keep himself in the loop and know the latest clinical gossips. Within the realm of critical care/cardiology, Dr. González-Hernández enjoys the topics of sepsis/septic shock, heart failure, and antithrombotic therapies.
I was going to try to find a good sci-fi snowy scene for here (think Star Wars), but then this popped up in my google search. And c’mon, there’s no doubt this punny bandit is the better option. (Image)
Ever since I heard about the concept of inducing hypothermia to a human being with the sole purpose of decreasing metabolism to preserve tissue and neurological function, I became fascinated by targeted temperature management (TTM). This seemed to be taken from a science fiction novel, and I was surprised this was actually a practice.
Perhaps you know TTM in your hospital as induced hypothermia, therapeutic hypothermia, targeted temperature management, and my favorite by far, Code Ice. (And it’s not my favorite just because Vanilla Ice’s hit song instantly populates my head and I hum it for the rest of my shift…)
Over the last 20 years or so, a few trials exploring and testing this concept have been released. The first of these trials was published back in the early 2000s, with another trial published in 2013, and more than a couple being released in the last 3 years. With the addition of several trials in 2021 (which btw, was the year of cardiac arrest with half a dozen clinical trials exploring different interventions in this setting), there certainly was a solid repository of journal club options for my students and residents.
So join me in exploring the basics of TTM, followed by a deep dive into the literature supporting and challenging this practice.
What is Code ICE?
I am sure many of us have been working in a hospital and heard a Code Ice being called on the overhead speakers. And if you were like me, you Immediately started wondering what in the world that meant. (Was it time for the ER staff to belt some "Let It Go”??)
Not exactly. Although that would be highly entertaining.
The first thing we need to ask ourselves is how does TTM work? The answer lies in knowing the ‘why’s’ for our actions. Knowing those ‘why’s’ enables us to fully understand concepts and rationales for the interventions performed nowadays in medicine.
This makes pharmacy practice less memorization and more thoughtful. So let’s start at the beginning.
During a cardiac arrest, a person’s body becomes deficient of oxygen. The longer a person is down, the higher the risk and incidence of damage to body tissue due to ischemia. Anoxic neurologic injury is an important cause of morbidity and mortality in these patients. If we recall from biochemistry and molecular biology, enzymatic reactions are largely mediated by temperature, and our body is chock full of enzymes. The higher the temperature, the faster these enzymes execute their reactions, with the opposite occurring at colder temperatures.
So connecting the dots, the main rationale of TTM is that lowering the body’s temperature slows down cellular metabolism, thereby limiting the damage caused by cerebral ischemia in the initial hours after a cardiac arrest.
Before diving into the literature, it’s important to note that TTM practice is not uniform across all institutions, and different institutions have variations among their protocols. In terms of inclusion/exclusion criteria, most institutions mimic the criteria implemented in the trials, which include but are not limited to:
Post cardiac arrest (any cause),
Return of spontaneous circulation (ROSC) < 30 mins from team arrival (some institutions have variations in time),
Time < 6 hours from ROSC (some places use 3 or 4 hours instead),
Patient is comatose or unconscious,
Mean arterial pressure (MAP) at least 65mmHg, and
No limitations to management in place .
Exclusions to TTM are often:
Advanced directive stipulating do not resuscitate (DNR),
Traumatic arrest,
Unwitnessed arrest,
Active bleeding,
Recent major surgery, and
Severe sepsis.
In most cases, patient criteria are very similar to these. Additionally, most institutions have the same overall procedure for TTM. However, as with many things, the devil is in the details, and TTM is no different. Most protocol variations between institutions relate to the methods used to induce hypothermia and how to manage a patient’s temperature. And the biggest beef? What the actual temperature goal for TTM is!
So TTM protocol variety really centers around two big questions:
What is the target temperature?
How do we get there?
(Image)
Looking first at the overall procedures for TTM, most protocols incorporate the following:
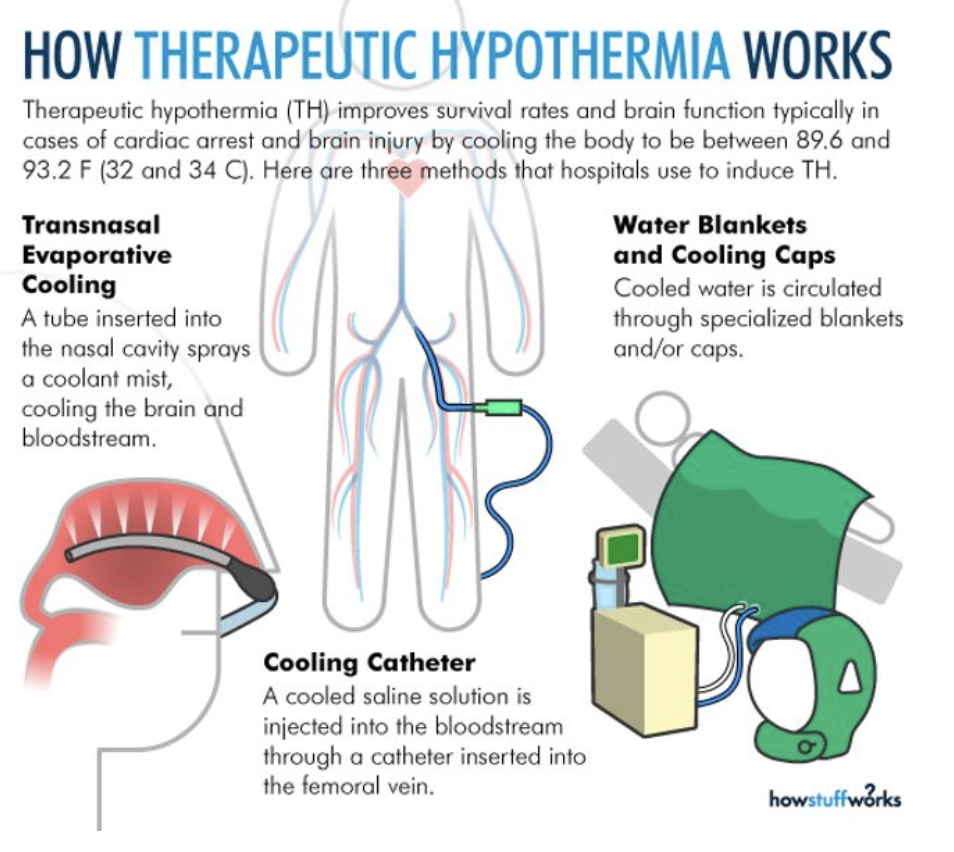
A Cooling Phase: Essentially the patient’s body temperature is lowered using a variety of methods. The three most commonly used methods include insertion of a cooling catheter, through which cooled IV fluids are infused. Other institutions use cooling blankets and/or cooling helmets, through which cooled water circulates, maintaining a reduced body temperature. And finally, there’s transnasal evaporative cooling. (My institution does not perform this last one routinely, but it is something I have read/heard about from colleagues at other institutions.)
During the cooling phase, the body temperature is reduced to our desired temperature over a 6-12 hour period. Most institutions use a temperature goal of 33C, while others use a goal closer to the 34C-36C range. There is some debate regarding the optimal target temperature, which of course is one reason we’re here at tl;dr today - to dive into the studies assessing the different temperature goals! So hang tight, we’ll get there.
Next is the Maintenance Phase: This is somewhat self explanatory… But once we have reached our targeted temperature goal, we work to keep the body temperature at that mark for 24 hours. (Other institutions might have this duration set to 12 hours, while others at 36 hours. But most places do it for 24 hours.)
Then we come to the Rewarming Phase: After reaching 24 (range 12-36) hours at our targeted temperature goal, the patient’s body temperature is rewarmed to 37C (98.6F) at a rate of 0.25C per hour.
Now, cooling the body below the temperature it was designed to function at is not an entirely benign process and can lead to complications. The first thing you might notice in a patient undergoing induced hypothermia is shivering, which is the body’s response to reset homeostasis. Although a natural response to the cold, this shivering can be counterproductive in the TTM setting since it causes the body’s temperature to increase. To keep this from happening, warming blankets might be used, along with optimizing sedation. If the previous modalities do not work, patients might undergo therapeutic paralysis with neuromuscular blockers.
Due to decreased metabolic demand during TTM, the body’s cardiac output also decreases. The factors that compose cardiac output, especially heart rate, are also decreased. Unfortunately, atrial fibrillation and other dysrhythmias are also possible, especially at lower temperatures.
During a cardiac arrest, oxygen becomes unavailable to many organs due to lack of perfusion, which leads to a shift to anaerobic metabolism and production of more lactic acid. Hypothermia affects the capability of the liver to metabolize this excess lactic acid, and metabolic acidosis develops. The triad of death is something we reference a lot in trauma, especially during massive transfusion protocols (MTP), because usually hypothermia, acidosis, and coagulopathy are associated with each other. (In the setting of trauma, the order of the equation is slightly different since the starting point is different, being coagulopathy. But the end result is the same.)
(Image)
So how does the triad fit here?
During cardiac arrest and TTM, we start with a lack of perfusion that leads to lack of oxygenation, an increase in lactic acid, and so on. When adding hypothermia to the equation, coagulopathies then become a possibility, especially when we are using lower target temperature goals. During hypothermia, as explained before, metabolic reactions are reduced. The coagulation cascade is essentially an enzymatic cascade, meaning this is largely dependent on temperature. If this is affected, the coagulation cascade is slowed down. However, the opposite is also possible since fibrinolysis and anticoagulation processes will also be slowed down. During TTM, many institutions avoid the use of anticoagulation to avoid affecting the process more than what is already affected by hypothermia.
Finally, electrolyte concentrations should be closely monitored during hypothermia. This is due to intracellular shifts that take place during hypothermia. During the Cooling Phase, potassium shifts from the extracellular space into the intracellular space, resulting in reduced serum potassium levels. However, during the Rewarming Phase, this shift reverses and rebound hyperkalemia is possible.
The main take away point is the following: during TTM, pursue a less aggressive potassium level between 3.0 - 3.5 mmol/L. Furthermore, when replacing potassium, it is good practice to not be as aggressive with potassium doses. A low hanging fruit in terms of interventions is to discontinue potassium replacement protocols in these patients, since these are mostly not tailored for this specific patient population.
Now that the basics of TTM (aka Code Ice aka Therapeutic Hypothermia aka Induced Hypothermia) are summarized, let’s dive into the evidence behind this practice. As a spoiler alert, to fully and accurately interpret the data, it’s a must to have some perspective about the trials that led to the 2021 TTM-2 trial.
A Deep Cold Dive into the Evidence for Targeted Temperature Management
The Bernard Trial
The trial that started it all was published by the Bernard group in 2002 in the New England Journal of Medicine. This study was performed in Australia. Patients in ventricular fibrillation when they arrived in an ambulance who were still comatose after ROSC and transferred to one of four participating centers were included in the study. Exclusion criteria were straightforward - men younger than 18 years or women younger than 50 years were excluded.
Now at first glance, this seems odd and disparate, right? Why were so many females excluded??
But the authors explained that this female age cutoff pretty much eliminated the possibility of any pregnant patients being included. So, essentially this criterion is a surrogate for pregnancy as an exclusion criteria, at the cost of excluding a large population of young women. Patients in cardiogenic shock were also excluded, defined in the trial as a systolic blood pressure of less than 90 mmHg despite epinephrine infusion. FInally, patients with other causes for coma outside of cardiac arrest, such as drug overdose, were excluded.
A word to the wise…
When looking at these trials it is important to remember the different interventions studied and the comparator group used. (Not all studies compared hypothermia versus normothermia, which is important to keep in mind moving forward.) The Bernard study…
Compared a temperature of 33C versus 37C,
Maintained hypothermia for 12 hours,
Rewarmed patients over 6 hours, and
Used cooling packs and paralytics to reduce the core body temperature and prevent shivering.
The primary outcome was survival to hospital discharge with sufficiently good neurologic function to be sent home or to a rehabilitation facility. As before, this is a straightforward, easily assessed endpoint that is patient-centered and clinically significant. Secondary outcome measures included the hemodynamic, biochemical, and hematologic effects of hypothermia.
A total of 77 patients were randomized, with randomization occurring based on the day of the week they were enrolled. Most baseline characteristics in regard to time to first shock, time to achievement of ROSC, number of shocks, and epinephrine dose administered were similar between groups. The biggest difference between groups was the percentage of patients who received bystander CPR in the normothermia group, which was significantly more than the hypothermia group.
The primary outcome was significantly higher in the hypothermia group, with 49% of patients achieving survival with enough neurological function to be discharged home or to rehab facility versus just 26% in standard care (p=0.046). Survival was not significantly different between groups (51% in hypothermia versus 68% in standard care, p=0.145).
However, it is worth noting that, to my knowledge, this study did not report the number of patients who had a fever during the duration of the intervention, which is an important piece of information that I will discuss in a bit. The Bernard study had additional limitations such as:
A close call, harrrr (Image)
Not recording baseline neurological function at ED arrival. If we do not know how these patients functioned at baseline, it’s hard to determine what kind of impact the intervention produced.
Lack of generalizability to younger women due to exclusion of women younger than 50 years.
The study was unblinded, which can lead to investigator bias. This refers to when investigators might consciously - or unconsciously - change their practices based on intervention awareness.
Small sample size. As a matter of fact, the fragility index (FI) of this study was only one. The Fragility Index (FI) is an important aid in the interpretation of clinical trial results. It measures how many events the statistical significance of a clinical trial result depends on. A smaller FI score indicates a more fragile, less statistically robust clinical trial result. In this study, a change of outcome in only one patient would have led to the results not being statistically significant.
Talk about a close call…
Although there were limitations, the Bernard trial was our first collection of evidence suggesting hypothermia might provide meaningful benefit in comatose patients after an out of hospital cardiac arrest.
Baby, It’s Cold Inside
The HACA trial
In the same 2002 NEJM issue that published the Bernard trial, there was also another related trial by another group of investigators. The Hypothermia After Cardiac Arrest (HACA) group trial was a randomized, controlled, multi-center, with blinded assessment of the outcome. This meant that the people taking care of the patients were not blinded, but the people assessing the neurological outcomes after 6 months were blinded.
Patients were included if they had a witnessed cardiac arrest with ventricular fibrillation or non-perfusing ventricular tachycardia (known today as pulseless ventricular tachycardia) with presumed cardiac origin of the arrest, an age of 18 to 75 years, a downtime of 5 to 15 minutes from the patient's collapse to the first attempt of resuscitation, and an interval of no more than 60 minutes from collapse to ROSC. (FYI, these criteria are the basis for a lot of subsequent trials and a lot of our protocols out there.)
Patients were excluded if they had any of the following:
Responsive after ROSC,
Prolonged hypotension after ROSC (at least 30 minutes at MAP < 60 mmHg),
Cardiac arrest after medical support already present,
Comatose prior to cardiac arrest due to drug overdose,
Hypoxemia (blood saturation less than 85 percent),
Pregnancy,
Terminal illness, or
Known coagulopathy.
Quite a list, but to this day, most of our protocols follow these criteria anyways.
If you haven’t run into this scale before, the CPC is a 5 item category scale with 1 being the best neurological outcome, meaning living a normal life, and 5 being the worst outcome, meaning death. (Image)
The HACA trial compared cooling patients to 32C-34C for 24 hours vs normothermia. The primary outcome was a favorable neurologic outcome within six months, defined as a Pittsburgh Cerebral-Performance Category of 1 (good recovery) or 2 (moderate disability). Mortality was recorded as a secondary outcome.
A total of 265 patients were randomized from a pool of about 3500 screened patients, suggesting some selection bias…which makes sense based on the long list of patient criteria. Baseline characteristics were similar between both groups.
The primary outcome of favorable neurologic outcome was significantly increased in the intervention group (55% of patients in the hypothermia group versus 39%, p=0.009). In terms of mortality, 6-month mortality was significantly reduced in the hypothermia group (41% versus 55%, p=0.02)
Now, let’s dig into some details regarding this trial. This protocol was slightly different from the one used in the Bernard trial. The methods to cool patients were the same, but the duration of induced hypothermia was 24 hours as opposed to 12 hours. The target temperature was arguably the same: 32-34C in this trial as opposed to 33C in the Bernard trial. This trial included more patients than the Bernard trial, which can count as a strength. Even though the people taking immediate care of the patients were not blinded to the treatment assignment, investigators assessing the neurological outcome at six months were blinded, which was intended to mitigate investigator bias.
On the other hand, this study had multiple limitations. Despite a bigger included sample size, over 90% of screened patients were excluded! This level of exclusion can limit the applicability of the study due to multiple population subtypes being left out. This is what we call selection bias in biostatistics.
Furthermore, the trial had to be stopped earlier than intended because of lack of enrollment, possibly a function of all the exclusion criteria used. This could have exaggerated the positive effect of the intervention. (Remember, the longer a trial is carried out, the more the results tend to regress to the mean.) Also, bias may have arisen because large random fluctuations of the estimated treatment effect can occur, particularly early in the progress of a trial.
Last, but not least, my biggest complaint about this study. The picture below presents the average temperature of participants per group. With lines representing the 75th percentile, this suggests multiple patients in the normothermia group might have actually experienced hyperthermia (aka fever = a temperature above 38C). This calls into question the findings of this study!
(Image)
Were the reported benefits of hypothermia observed in this study because of the cooler temperatures, or did hypothermia look so good because so many people in the normothermia group actually experienced fevers, which could be harmful after a cardiac arrest?
Despite this big caveat, the HACA trial presented a number needed to treat of 6, which is quite impressive. Meaning that for every 6 patients treated with induced hypothermia, 1 patient would have a neurological benefit at 6 months. Based on this and the Bernard trial, induced hypothermia to a goal temperature of 32-34C was implemented in multiple institutions across the globe.
Now that we have some background, it’s time for the drama. This whole TTM strategy has recently been called into question with the release of the TTM and TTM2 trials.
Groundhog Day for Induced Hypothermia
The TTM Trial
This is where the initial paradigm shift occurred. No large trials assessing the utility of induced hypothermia took place for nearly a decade until Nielsen and colleagues published the TTM trial in 2013. This was a multicenter, single blind, randomized controlled clinical trial across 36 ICUs in Europe and Australia. This trial compared targeted temperature strategies of 33C versus 36C for 28 hours. Protocols for both temperature groups provided for active avoidance of pyrexia for 72 hours. Therefore, both groups’ strategies differed slightly from previous trials with the active avoidance of fever for 72 hours.
Patients were included if they were:
At least 18 years of age,
Unconscious after cardiac arrest (a score of <8 on the Glasgow Coma Scale [on which scores range from 3 to 15, with lower scores indicating reduced levels of consciousness]),
Presumed cardiac arrest of cardiac source, and
Sustained ROSC for at least 20 minutes.
On the other hand, some exclusion criteria are listed below:
Pregnancy,
Known bleeding diathesis (other than medically-induced coagulopathy, e.g., warfarin),
Suspected or confirmed acute intracranial bleeding or acute stroke,
Unwitnessed cardiac arrest with initial rhythm asystole,
Do Not Resuscitate (DNR) order,
Terminal illness with 180 days survival unlikely,
Known pre-arrest Cerebral Performance Category 3 or 4,
>4 hours from ROSC to screening,
SBP <80 mmHg despite fluid loading/vasopressor and/or inotropic medication/intra-aortic balloon pump, or
Temperature on admission <30°C.
It is important to note that initial rhythm was not part of the 2013 TTM trial’s inclusion criteria, which is a big difference from the other trials that only included shockable rhythms (Vfib or pVT). Hypothermia was achieved via ice packs, cold blankets, cold IV fluids, and using sedation and paralysis. Prevention of fever in the 72 hours post TTM was left to the discretion of the study sites. At that time, a neurological evaluation by a blinded person was performed, and this person recommended whether or not to continue treatment or withdraw care.
The primary outcome was all-cause mortality through the end of the trial with a mean follow-up of 256 days. The main secondary outcome was a composite of poor neurologic function or death, defined by Cerebral Performance Category (CPC). (For simplicity’s sake, this was the same scale used in the primary outcome of the HACA group trial.)
Other secondary outcomes were the CPC at discharge from the ICU and from the hospital, as well as the best (aka the numerically lowest) reported CPC during the trial period. Predefined serious adverse events were recorded up to day 7 in the ICU.
A sample of 900 patients would have provided 90% power to detect a 20% reduction in the hazard ratio for death. A total of 950 patients were enrolled between November 2010 and January 2013. Of these patients, 476 were randomly assigned to the 33C group and 474 to the 36C group. Baseline characteristics were similar in both arms, with males being 80% of the patients. Most events were witnessed and occurred at home, with a bystander performing CPR (73% in both arms…go bystanders!!).
Almost 80% of patients had a shockable rhythm, although 11% were in asystole and 6-8% were in pulseless electrical activity. This is important because previous trials only included patients with shockable rhythms, i.e., vfib or pVT. There was no difference in the start of BLS, the start of ACLS, and achievement of ROSC (1 minute, 10 minutes, and 25 minutes, respectively).
When looking at the temperature curves, we observe that a lesser proportion of patients had fever. The colored bars represent 2 standard deviations (95th percentile), as opposed to 75th percentile in the HACA group trial. Even capturing more patients, fewer patients in the higher temperature group experienced temperatures of >38C.
There was no difference in all-cause mortality through the end of the trial. Forty-eight percent of patients died in the 36C group, and 50% died in the 33C group. The hazard ratio for death was 1.06 for cooling to 33°C (CI 0.89 to 1.28; p=0.51). The groups did not differ significantly with respect to the composite outcome of death or poor neurologic function at 180 days (risk ratio for a CPC of 3 to 5 in the 33C group, CI 0.88 to 1.16; p=0.78). In terms of side effects, there was no difference between groups except for electrolyte disturbances, which were seen more in the 33C group.
This trial was the biggest TTM investigation yet, including over 900 patients. Plus, in terms of methodology, this trial has been critically acclaimed. It is worth noting that health care professionals caring for the trial patients were aware of the intervention assignments. (I mean, it is almost impossible to blind something that is measured multiple times per shift in hospitalized patients.)
But the physicians performing neurologic prognostication, the assessors of neurologic follow-up and final outcome, the study administrators, the statisticians, and the authors were unaware of the intervention assignments. They even remained unaware of intervention assignment until after the manuscript was submitted, which is a heck of an effort to reduce investigator bias. Furthermore, the primary outcome of death is basically as objective as you can get and cannot be changed by lack of blinding.
The TTM trial has more applicability than previous studies since all different types of rhythms were included. Additionally, more than 70% of screened patients were included in the trial! The study was robustly powered at 90%, which is even more than the standard accepted 80%, so the lack of difference in the primary outcome shouldn’t have been due to a type II error (aka incorrectly accepting the null hypothesis that there was no difference).
However, the TTM study was powered to detect a RRR of 20% or an ARR of ~11%, so it’s possible that a smaller treatment effect remained undetected. So why was that treatment effect size chosen when determining power and target enrollment? When looking back at the previous studies, the HACA group trial found an ARR in death of 14%. So this is likely where that number might have been pulled. It’s not as random as it may initially seem…
Of note, this trial is acknowledged avoidance of fever as a clinically important goal. And it really did try to answer the question of TTM benefits; however, given both groups had a protocol in place to avoid fever for 72 hours, it is hard to know what actually conferred benefit. Was it the avoidance of fever or having a TTM protocol in place?
Well darn.
But thanks to this trial, what we do know is that avoidance of fever combined with TTM to 36C might be beneficial. So this trial gave us the first piece of evidence that perhaps we don’t need to be quite so aggressive when treating these patients.
The Cold TTM War
The HYPERION Trial
There were some other trials were published in between the TTM and the TTM2 trial; however, the 2019 HYPERION trial was probably the most discussed one from this time period. HYPERION was an investigator-initiated, open-label, blinded-outcome-assessor, pragmatic, multicenter, randomized, controlled trial in 25 intensive care units (ICUs) in France. The trial compared moderate therapeutic hypothermia (33°C during the first 24 hours) with targeted normothermia (37°C) in patients resuscitated after non-shockable rhythms. (For context, remember that the TTM trial was the only trial we’ve discussed thus far to include non-shockable rhythms.)
Inclusion criteria for HYPERION were as follows:
Age at least 18 years,
Resuscitated from out-of-hospital or in-hospital cardiac arrest with a non-shockable rhythm due to any cause (i.e., these weren’t just cardiac patients like other trials to date), and
GCS 8 or less at ICU admission (or if sedated, GCS score prior to sedation).
Exclusion criteria were:
No-flow time (aka time from collapse to initiation of CPR) > 10 minutes, meaning no CPR within 10 minutes of collapse,
Low-flow time (aka time from initiation of CPR to ROSC) > 60 minutes,
Epinephrine or norepinephrine infusion > 1 mcg/kg/min,
Time from cardiac arrest to screening >300 minutes,
Moribund (there’s a good SAT word for you),
Child-Pugh Class C cirrhosis of liver, or
Pregnant or breastfeeding.
Each center followed its standard protocol (active internal cooling with a specific device, active external cooling with a specific device, or active external cooling without a specific device). Rewarming was then performed at a rate of 0.25 to 0.50C per hour, to 36.5 to 37.5C, which was maintained for 24 hours.
The primary outcome was the proportion of patients with a CPC of 1 or 2 at day 90, so mostly a neurological function outcome, rather than mortality. (This was more in line with the primary outcomes of the HACA and Bernard group trials.) The CPC score at 90 days was assessed during a semi-structured telephone interview. The secondary outcomes were mortality, mechanical ventilation duration, length of stay in the ICU and hospital, infections, and hematologic adverse events.
The investigators assumed that 23% of the patients in the hypothermia group and 14% of those in the normothermia group would have a CPC score of 1 or 2 on day 90. Using these numbers, they determined that 584 patients would be needed to be enrolled for the trial to achieve a power of 80% to detect an ARR of 9% in the primary outcome.
In the end, a total of 581 patients were enrolled, meaning the trial did not reach power. (We’ll find out whether this matters momentarily…). Most characteristics were similar between groups. A few characteristics that stand out are the following:
In-hospital cardiac arrest: 26% versus 29% (Hypothermia vs Normothermia, respectively)
Bystander witnessed cardiac arrest: 97% versus 92%
Bystander CPR: 70% in both groups
In terms of cause of cardiac arrest, ~55% were from asphyxia, and a little over 25% were from cardiac causes.
Regarding the primary outcome, 29 had a CPC score of 1 or 2, as compared with 17 in the normothermia group (10.2% vs. 5.7%; difference, 4.5 percentage points; 95% confidence interval [CI], 0.1 to 8.9; P=0.04). No significant difference in 90-day mortality was observed: 81.3% of patients died in the hypothermia versus 83.2% in the normothermia group (no reported p value).
The HYPERION study had some strengths. It was randomized, multicenter, and included patients with in-hospital cardiac arrest. It focused on non-shockable rhythms, as well as events from non-cardiac sources.
BUT it also had many, maaaany limitations!
No data was provided regarding time to CPR, ACLS, or ROSC. Variations in these factors could have impacted the likelihood of patient success, regardless of TTM. Perhaps the most glaring and important limitation is that there were some methodological differences between the groups. More than 5% of patients had a temperature above 38C in the first 48 hours, which qualifies as hyperthermia.
There is a verrrry big difference between RASS of 0 and -5. What did that mean for patients in HYPERION? (Image)
Furthermore, patients in the hypothermia group received sedation to a RASS of -5 until they were rewarmed. Meanwhile, in the normothermia group, patients received sedation to a goal RASS of 0 for a duration of 12 hours. Other trials have used similar sedation strategies across both groups. This is important because patients are sedated regularly in the setting of cardiac arrest, which aids in achieving a desired temperature goal and preserving neurological function.
Another methodological difference was the time taken to decide about withdrawal of care, which occurred sooner in the normothermia group. Lastly, methodological differences between groups now also calls into question the study intervention’s effect since we have added a confounder to the equation.
Other limitations include:
Possible recall bias. Primary outcome was assessed with telephone interview, which is highly dependent on the patient’s (or patient's next of kin) subjective point of view. Recall bias is a systematic source of bias likely to occur when patients do not remember certain events or think certain events might not be important. When assessing neurological function, this could play a big role.
Some important side effects of hypothermia were not reported, including incidence of arrhythmias or electrolyte disturbances.
Trial was only performed in 1 country, limiting applicability. A number of patients had their sedation stopped early due to suspected brain death, which is highly dependent on local laws and local practices.
Low fragility index (just one!). This is less than the number of patients that withdrew from the study and also less than the number of patients with missing data. So what would have happened to the results if the patients didn’t withdraw or had all their data?!
This study did not meet power. Remember that only 581 people were included rather than the desired 584? But wait, is this actually a limitation? A significant difference was detected in the primary outcome, so the fact that the study didn’t meet the desired enrollment isn’t necessarily the end of the world. This would have mattered if no difference was found between groups because then perhaps we could be dealing with a type II error (and therefore potentially a missed treatment effect).
But because there was a difference detected, those extra 3 patients don’t really make or break the statistics here. However, we still have to consider the other pieces of the methodology to determine if we accept the study results or not… The story doesn’t stop at a “statistically significant” primary outcome. (I’m talking about the protocol differences between groups, whether or not you believe the power and sample size calculations were clinically appropriate, and that fragility index of 1!)
(Image)
All this to say, the HYPERION study is one of those trials I like to present to my students and residents to remind them that not everything published is of the highest quality. There were several sources of bias that very well could have affected the study results.
Will TTM Ever Be Cool Again?
The TTM-2 Trial
This is the newest kid on the TTM block. The TTM-2 trial was just published in the New England Journal of Medicine in 2021. It compared hypothermia at 33C for over 28 hours versus targeted normothermia 37.5C. (If a patient’s temperature reached 37.8C, a cooling device was used to return to 37.5C.) This was a multicenter, international, randomized controlled trial across 14 countries and 61 institutions.
The FOUR score, if you haven’t run across this one. (Image)
Some inclusion criteria were:
Out-of-hospital cardiac arrest of presumed cardiac or unknown cause without any limitation to rhythm,
Sustained ROSC: >20 minutes of circulation without need for chest compressions,
Unconscious: as defined by the FOUR score motor response and not able to obey verbal commands. (This scale has 4 different domains that range from 0 to 4, and these scores are added together. This trial included patients with a total score of <4.)
No limitations to management (i.e., no DNR), and
Randomization must have been no later than 180 minutes after ROSC.
On the flip side, patients were excluded if they had any of the following:
Unwitnessed cardiac arrest with initial rhythm asystole,
Temperature on admission < 30C,
On ECMO prior to ROSC,
Suspected pregnancy,
Intracranial bleed, or
Severe COPD with long-term home oxygen therapy.
With regards to how TTM-2 was executed, sedation was mandated in both groups until the end of the intervention period. Both groups had sedation titrated to a RASS of -4. After the intervention period, a normothermic target (36.5C to 37.7C) was maintained until 72 hours after randomization in patients who remained sedated or comatose.
As with the other trials, healthcare workers taking immediate care of the patients could not be blinded; however, investigators and those who assessed neurological function were blinded. Furthermore, two different manuscripts were written to make sure investigators remained blinded with regards to the outcome.
The primary outcome was all-cause mortality at 6 months. The main secondary outcome was the proportion of people with a poor functional outcome (modified Rankin scale, mRS 4-6) at 6 months. This scale goes from 0 to 6 with 0 representing no symptoms and a score of 4 representing inability to walk without assistance. A score of 5 represents bedridden, and 6 represents death. These outcomes were similar to the original TTM trial, with the only difference being the scale used to assess neurological outcomes. Adverse events were also recorded in this trial, similar to TTM.
The investigators estimated a sample of 1862 patients would be needed to provide 90% power to detect a relative reduction of 15% in the risk of death (~ARR of 7.5%). This was rounded to 1900 to account for loss to follow up and other variables. 4355 patients were screened, of which 1900 were enrolled. Thirty-nine of those patients withdrew or didn’t provide consent, which left 1861 patients for randomization. (So close…)
Both patient groups had similar incidences of comorbidities. Over 90% of patients in each group had a witnessed cardiac arrest with 82% of patients in the hypothermia group and 78% of patients in the normothermia group receiving CPR from bystanders. (Seriously, these bystanders are killing it with jumping into action!) Nearly 75% of patients in each group had a shockable rhythm, with each group achieving ROSC by 25 minutes on average. All patients had similar rates of cardiovascular interventions in each group.
There was no difference in the primary outcome of death at 6 months. Fifty percent (465/925) in the hypothermia group versus 48% (446/925) in the normothermia group passed, RR 1.04 (95% CI 0.94-1.14, p = 0.37). It is worth noting that 11 patients were lost to follow up; therefore, a total of 1850 patients were included in the analysis. Several prespecified subgroup analyses were performed for the primary outcome, and the results were consistent across gender, age, time to ROSC, and type of initial rhythm.
For the secondary outcome of proportion of people with a poor functional outcome (modified Rankin scale, mRS 4-6) at 6 months… There was no numerical nor statistical difference between groups, 55% vs 55% (RR 1.00, 95% CI 0.92 – 1.09). For both outcomes, a best-worst case scenario analysis was performed. This is done to ensure that the outcomes weren’t altered because of patients being lost to follow up. In this case, there was no difference.
In terms of side effects, arrhythmias resulting in hemodynamic compromise were more common in the hypothermia group than in the normothermia group (24% versus 17%; p<0.001).
Now, let’s move to the supplementary appendix, because in this day and age, you can find a lot of juice in those appendixes. When we look at the proportion of patients that had life support withdrawn, it is similar between groups. This is opposed to the HYPERION trial, in which these decisions were more common in the normothermia group early on. This is in part because the TTM-2 investigators had a protocol in place to handle this, which is one of the strengths of this study.
Btw, the y axis scaling on this graph drives me batty. Whyyy so much white space?? I guess that’s what happens when you’re a graph that only makes it to the supplementary appendix rather than the main publication… (Image)
The investigators also reported the proportion of patients who developed fever in the study. Fever incidence was slightly higher in the normothermia group in the first 40 hours after randomization. The biggest difference between this trial and other trials is the fact that, once patients reached the 37.7C mark, cooling took place. All in all, about 46% of patients in the normothermia group had cooling performed to a temperature of 37.5C via similar means that those in the hypothermia group (cooling devices, cold IV fluids, etc).
This trial has been praised for its solid internal validity. Only 3 patients in total deviated from their protocol, and protocols for sedation and withdrawal of life support were put in place to avoid the variations in protocol seen in the HYPERION trial. To date, this is the largest trial studying the topic of TTM after cardiac arrest, and it included almost 1000 more patients than the original TTM trial.
The TTM-2 trial also did a good job including other types of rhythms (not only shockable rhythms like the original studies). Blinding of investigators was performed in a similar way to the original TTM trial, even creating a fake manuscript to avoid the investigators from looking at the data and trying to extrapolate from it. So in that regard, the investigators deserve praise because several tools were used to limit sources of bias. Lastly, the inclusion of pre-specified subgroup analyses tried to answer some questions, such as whether hypothermia might be a better option with those with a time to ROSC over 25 minutes.
The biggest limitation of this trial is the fact that in-hospital cardiac arrest patients were not included, limiting applicability to this group, a group that has been excluded from most trials on this topic. Nearly 50% of screened patients were not included, which hints at selection bias…but at least, an explanation as to why most of these patients were excluded was provided. A potential confounder was that 20% of patients were co-enrolled in the TAME trial, 10% in each group. This could potentially be a confounder, but we will only be able to determine this once the TAME trial is published. Lastly, in the power calculation, the authors used an ARR of 7.5% in mortality, which might have been aggressive when looking at the results of the original TTM trial. The investigators used the ARR from previous trials (HACA and Bernard).
All in all, the TTM-2 is an important trial that seems to reinforce the idea that perhaps we do not need to be super aggressive when performing TTM in our patients. To further this idea, later in 2021 after TTM-2, another smaller trial was published. The CAPITAL CHILL trial, which compared 31C versus 34C, was unable to find any difference in the primary outcome of all-cause mortality and poor neurological outcome at 180 days.
Guideline TTM Recommendations
The ACC/AHA and Europe have similar stances on TTM. Of note, the ACC/AHA guidelines were published in 2020, and the European guidelines in early 2021 - both prior to the publication of the results of the TTM-2 trial.
Both the European and ACC/AHA guidelines recommend TTM for adults after either out-of-hospital cardiac arrest (OHCA) or in-hospital cardiac arrest (IHCA), irrespective of rhythm, as long as they remain unconscious after ROSC. The guidelines provide a wide range for goal temperature, suggesting a goal temperature of 32C to 36C, with avoidance of fever in the next 72 hours.
The guidelines comment that, to date, no trial has compared controlled normothermia versus no fever control. Their rationale for recommending TTM and providing such a large goal temperature range is the theoretical benefits of cooling patients after cardiac arrest along with the evidence from the Bernard trial, the HACA trial, and the HYPERION trial, where benefits were seen with the implementation of TTM with a goal between 32C and 34C. The TTM trial was the biggest study to date, but only compares 33C vs 36C. Hence, the panel decided to include 36C in their range due to the strength of the TTM study and the lack of a difference between the 2 temperature groups.
The tl;dr on Targeted Temperature Management (TTM)
As mentioned before, perspective - specifically, historical perspective - is important. This is a prime example of why decisions should be made by looking at the bigger picture and not just at a single study. Even though there are guidelines in place, some important questions remain.
What is it that confers benefits? Is it the avoidance of fever or having a TTM protocol in place?
At this point, based on the TTM-2 trial, it seems that avoidance of fever is what confers the most benefit. The only trials to show TTM benefit are those that allowed patients in the normothermia group to develop fever. A recent trial comparing 32C vs 34C, showed no benefit between groups. Now, the real question in my eyes is this: do we need to target 36C or controlled normothermia? If we ever get a TTM-3 trial, I hope it tries to answer this question.
Time to CPR in the original TTM trial seemed surprisingly low (1 minute). Is a lower temperature TTM goal more beneficial in patients with more severe anoxic brain injury?
The TTM-2 trial had a subgroup analysis that showed that patients with a ROSC >25 minutes after CPR did not benefit from a lower temperature goal of 33C compared to aggressive normothermia. We do not have dedicated trials assessing this, but the available data seems to suggest a lower temperature may not provide additional benefits in these patients.
Every TTM trial to date has used different TTM goals and different times in goal therapy. What is the optimal duration of therapy and optimal temperature goal?
At this time, we have studies comparing hypothermia (32-34C) vs normothermia (with some patients developing fever). We have a trial comparing 33C versus 36C (TTM). The TTM-2 trial studied 33C versus controlled normothermia. But no trials have studied 36C versus normothermia. Nor have any studied normothermia with prevention of fever versus no prevention of fever.
But to be honest, due to what we know (that fever is harmful in these patients), I do not think that will ever happen from an ethical standpoint. We might one day have a trial of 36C vs normothermia, which I would be very excited about!
The available evidence, taking into account the six main trials, seems to suggest that avoiding fever is the most important intervention that we can do after a cardiac arrest. The only trials to show a benefit with lower temperatures allowed patients to develop fever (HACA and HYPERION). The two strongest trials to date (the TTM and TTM-2) compared less aggressive goals to the aggressive goal of 33C, and no difference was seen. Both of these trials made honest efforts to avoid fever in their patients and were methodologically superior to the other trials published in the same topic, along with robust sample sizes.
So based on what we have, even after the release of the guidelines, I think the evidence leans toward avoiding fever (aka targeting aggressive normothermia). Now…whether we need to pursue 36C or normothermia remains unanswered. Most providers I work with nowadays lean toward pursuing a goal of 36C, and I do agree with this.
(Image)
We know that lower targets are associated with electrolyte disturbances and arrhythmias, and with COVID-19 still in our lives, staffing has been an ongoing issue. TTM is mostly nurse-driven and needs almost a 1:1 staffing ratio to make it happen, especially in those cases where a more aggressive goal is pursued. If we have an evidence-based intervention that produces similar results, with less side effects, and less resources employed, I am a firm believer that that intervention is what makes the most sense.
I do think we are in the middle of a paradigm shift and future guidelines will be leaning more towards a less aggressive goal, based on the results of the TTM and TTM-2 trials. In the meantime, a goal of 36C to aggressive normothermia is what I will try to implement in most of my patients in the near future.